Chapter 3:
Consequence
Significant consequences were obtained in order to qualify the ERK isoforms in MDA-MB 231 chest malignant neoplastic disease cell lines by several experiments.
3.1. IC50 value of Doxorubicin in MDA-MB 231:
IC50 value of Doxorubicin investigated for MDA-MB 231 was 3200nm.
3.2. Morphology of Normal MDA-MB 231Cells:
MDA-MB 231 cells are epithelial cells, which form spindle form constructions and have compact morphology.

Figure 3.1:Normal MDA-MB 231 ;
- 80 % feeder cells( B )Full feeder cells.
3.3. Morphologic Analysis of Drug influenced MDA-MB 231 cells:
A chest malignant neoplastic disease cell line MDA-MB 231 was used to do drug immune cells. Two chemotherapeutic drugs, doxorubicin and paclitaxel were utilized in a considerable scope of concentrations in separate experiments with MDA-MB 231 cells. During the development of cellular opposition to these both drugs, many morphological alterations were detected in MDA-MB 231.
3.3.1. Affect of Doxorubicin on MDA-MB 231 Cells:
MDA-MB 231 cells were treated hebdomadal with different doses of doxorubicin to bring on opposition and this lead to morphological transmutations in normal MDA-MB 231 cells. Cells formed extended constructions upon exposure to the drug and apoptotic organic structures were besides examined under microscope as the doxorubicin concentrations were increased.

Figure 3.2:Different cell morphologies of MDA-MB 231after intervention with doses of doxorubicin ;( a )3nm,( B )4.5nm,( degree Celsius )6.7nm,( vitamin D )13.4nm,( vitamin E )10.05nm,( degree Fahrenheit )15.1nm,
( g )22.5nm,( H )33.7nm.
3.3.1a. Tolerable dosage of Doxorubicin:
MDA-MB 231 was treated to the maximal concentration of doxorubicin until tolerable dosage was achieved. Conc. 33.7nm was the highest and tolerable dosage of the drug on MDA-MB 231.
3.3.1b. Doxorubicin immune Cell:
After the 15 hebdomads of doxorubicin intervention on MDA-MB 231, chemoresitant cells were obtained at 22.5nm conc.
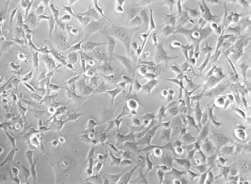
Figure 3.3:Drug immune MDA-MB 231.
3.3.2. Ctytoxic consequence of Paclitaxel:
Cytotxic drug paclitaxel induced opposition in MDA-MB 231 at really low concentration compared to doxorubicin. And some morphological changes besides examined in this procedure.

Figure 3.4:Paclitaxel treated MDA-MB 231 cells ;( a )0.56pm,( B )1.67pm,( degree Celsius )5.0pm,( vitamin D )15.0pm,( vitamin E )375pm,( degree Fahrenheit )1.8nm,( g )3.5nm,( H )7nm.
3.3.2a. Highest bound of Paclitaxel Dose:
MDA-MB 231 were maximally treated to the 7nm of paclitaxel, dose 9 that was least tolerated by the cells.
3.3.2b. Concentration of Immune Cells:
Paclitaxel opposition was induced in MDA-MB 231cells after handling them for the clip period of 15 hebdomads and cells became immune at dose 8 i.e. , 3.5nm conc.
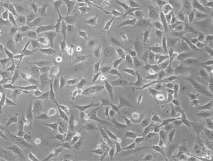
Figure 3.5:Paclitaxel immune cells.
3.4. Evaluation of duplicating clip of Normal and Resistant MDA-MB231:
Experimental informations of duplicating clip and proliferation of normal and both doxorubicin and paclitaxel immune MDA-MB 231 was evaluated at 0 hour, 24hr, 48hr and 72hr.

Figure 3.6: Doubling clip informations of MDA-MB 231.
3.5. Curative response of ERK 1/2 Inhibitor:
ERK activity was targeted in normal and both doxorubicin and paclitaxel immune MDA-MB 231 by presenting ERK1/2 inhibitor and cell viability was assessed with the MTT check.
3.5.1a. Data of Normal MDA-MB 231:
Inhibitor treated informations of normal cells was studied at 0hr, 24hr, 48hr and 72hr.

Figure 3.7:Data of inhibitor treated normal MDA-MB 231 with control DMSO and untreated control cells.
3.5.1b. Datas of Doxo-resistant MDA-MB 231:
Data of inhibitor treated and untreated Doxo-resistant cells were collected at 0hr, 24hr, 48hr and 72hr.

Figure3.8:Data of inhibitor treated Doxo-resistant MDA-MB 231 with control DMSO and untreated control cells.
3.5.1c. Datas of Paclitaxel-resistant MDA-MB 231:
Data of inhibitor treated and untreated paclitaxel-resistant cells were collected at 0hr, 24he, 48hr and 72hr.

Figure3.9:Data of inhibitor treated Paclitaxel-resistant MDA-MB 231 with control DMSO and untreated control cells.
3.6. Competence of Colony formation:
Colony organizing check was conducted on both normal and drug immune MDA-MB 231 but merely normal cells formed settlements.

Figure 3.10:Clonogenic check of normal MDA-MB 231 ;( a )Untreated control,
( B )Inhibitor treated normal cells.
Chapter 4
Discussion
Breast malignant neoplastic disease is the malignance of chest tissues which is distributing quickly in the universe and besides the taking cause of malignant neoplastic disease decease in adult females ( Falandry, Canney, Freyer, & A ; Dirix, 2009 ) . Basicss of malignant neoplastic disease patterned advance in chest mammalian epithelial cells are assorted mutated cellular events including cell growing and other normal cell endurance and cell decease related activities controlled by signaling Cascadess, that eventually leads to untypical and hyper-active invasive malignant neoplastic disease cells ( Albertson et al. , 2003 ; Jeffrey et al. , 2005 ) . These signaling Cascadess comprise of protein kinase signal transduction tracts in which ERK/MAPK pathways is most outstanding ( Brantley-Finley et Al, 2003 ; Sebolt-Leopold, 2000 ) .
Extracellular signal-regulated protein kinase ( ERK ) Cascadess regulated by ERK isoforms ( ERK 1, 2, 3 and 5 ) involve in sub-cellular signaling and monitoring of its bound maps like cell endurance and cell decease ( Santen et al. , 2002 ) . Garnett and Marais ( 2004 ) described in their survey that ERK pathway chiefly promotes cell proliferation and transmutation of normal cells to malignant in chest tissues. They illustrated that the strength of this ERK activity in chest cancerous tissue besides gives information about the activation degree of other growing factor stimulated tracts. Largely, ERK1/2 tracts are reported with hyperactivated ERK 1 and 2 in the elevated proportion of immortal cells in cancerous mammalian tissues ( Platanias, 2003 ; Santen et al. , 2002 ) . Alongwith ERK 1/2, isoforms ERK 5 besides a chief regulator of cancerous growing, but its elaborate map in ternary negative chest malignant neoplastic disease is still unknown ( Esparis-Ogando et al. , 2002 ; Weldon et al. , 2002 ) . That is why, ERK isoforms can be of import factors to analyze cell’s hyperactivity and root cause of cancerous transmutations ( Taherian, & A ; Mazoochi, 2012 ) . Furthermore, their word picture can back up old researches to aim kinases signaling Cascadess for curative intercessions and they can be projected as a malignant neoplastic disease forecaster and clinical predictive marker.
To analyze and uncover the mechanism of signal trafficking inside the cell and ultimate cell destiny, different cell lines of chest carcinoma e.g. , MDA-MB 231 are of great importance because the cell lines offer an unrestrained beginning of information for research to understand cell XT between cells in the human organic structure. And by developing chemoresistance in malignant neoplastic disease cells lines, it helps to expose the heterogeneousness of such cells.
This current survey was conducted on chest malignant neoplastic disease line MDA-MB 231 to qualify the extracellular signal-regulated protein kinase ( ERK ) isoforms. The chief focal point of the research was to detect the sub-cellular signaling by ERK isoforms 1, 2, 3 and 5 inside chest malignant neoplastic disease cells through which cell proliferation, cell endurance and apoptotic procedures are regulated. This purpose was achieved by set uping chemo-resistant MDA-MB 231cells against two different drugs, viz. doxorubicin and paclitaxel and so differences occurred due to drug influence on their proliferation rates and programmed cell death were examined.
IC 50 ( repressive concentration to bring forth 50 % cell decease ) value of the doxorubicin in MDA-MB 231 was 3.2um ( 3200nm ) estimated which is really high as compared to earlier reported IC 50 of Doxo-treated MDA-MB 231 by Taherian et al. , ( 2012 ) . The ground behind this might be the hapless quality and less specific activity of the used doxorubicin in the lab.
Normal cells of MDA-MB 231 were observed really compact and spindle form in construction, but debut of both chemotherapeutic deformed the normal morphology of cells in order to do immune cells. For this intent, hebdomadal doses of doxorubicin and of paclitaxel were set.
10 doses of doxorubicin were selected from the concentration of 3nm ( 1000 fold less than IC50 ) to 33.7nm ( tolerance bound ) and merely 1.5 fold drug conc. was increased in each dosage. After dose 4 ( conc. 6.7nm ) , 2 fold drug conc. ( conc. 13.4nm ) was increased, but cells could non survived and conc. was reduced to last conc. Therefore, old tendency was followed to choose the drug dosage. At get downing concentrations, cells easy started fring their compact and normal morphology and became more extended and idiot as the conc. of drug increased from low to high. In the starting of the hebdomads, more cell decease was observed, but in the terminal, cells were observed more stable towards applied conc. of drug. Dose 10 ( conc. 33.7nm ) was examined most tolerated by the cells. About 15 hebdomads of intervention resulted in Doxo-resistant MDA-MB 231 at 22.5nm because at conc. 33.7nm, cells wholly died after one hebdomad.
Same tendency was followed to do paclitaxel immune cells. 9 doses were selected from 0.56pm to 7nm. Different creases of conc. were set after analysing the conditions of cells. Compared to Doxo-resistant cells, morphological alterations were detected in lower conc. in paclitaxel influenced cells. Dose 9 ( conc. 7nm ) was the tolerance conc. on the cells. Immune cells were achieved at 3.5nm conc. ( dose 8 ) . This was examined in both chemo-resistant cells that they about attained MDA-MB 231 natural morphology.
Proliferation and growing rate of normal MDA-MB 231 and both drug resistant cells was analyzed by MTT assay at different clip intervals. Data shows that after 24 hour, normal cells proliferated more quickly than immune cells and rate were besides high in paclitaxel-resistant cells than Doxo. After 48 hour, same tendency was observed, but surprisingly, informations evaluated after 72 hour shows growing increase in Doxo-treated cells but decrease in normal and paclitaxel treated cells. This information shows that proliferation ability is greatly affected in Doxo-treated cells compared to paclitaxel affected cells and besides took some clip ab initio to get the better of the emphasis of sub-culturing but after that started turning easy.
ERK 1/2 are the most outstanding ERK isoforms in chest malignant neoplastic disease growing as described above. Therefore, Specific inhibitor of ERK 1/2, FR180204, was selected to aim these both isoforms in MDA-MB 231 with greater selectivity. This inhibitor has alone interactions with ERK isoforms at the molecular degree. It interacts with the typical residues of ERK1/2 and performs its repressive action on mark ( Ohori et al. , 2005 ) . The aim of this survey was besides to find the activity of ERK isoforms particularly ERK 1/2 in chest malignant neoplastic disease. So, inhibitor check was carried out to verify whether the normal, Doxo and paclitaxel treated MDA-MB 231 show reduced cell growing and proliferation.
Normal cells were treated with inhibitor and cell viability informations was recorded after 0hr, 24hr, 48hr and 72hr with MTT at 420nm. After 24 hour, inhibitor treated cells shows inhibited growing as compared to command but after 72 hour, information shows more growing in targeted cells than control. This may be due to acquired opposition of cells against ERK 1/2 inhibitor.
Datas from Doxo and paclitaxel was besides inspected in the same mode and both drug resistant cells show reduced cell growing and cell viability than control untreated cells. But interestingly, assay informations for normal and immune cells is demoing less growing in DMSO control which is due to unknown ground.
To prove the competence of normal and immune MDA-MB 231, settlement organizing check was performed. Cells were cultured and treated with inhibitor and left for 14 yearss so, settlements were fixed and stained with glutaraldehyde/crystal violet. Merely normal MDA-MB 231 control and inhibitor treated cells formed settlements, as shown in figure 3.10.





